SHISEIDO FUTURE SOLUTION LX DAYTIME PROTECTIVE
SHISEIDO FUTURE SOLUTION LX DAYTIME PROTECTIVE
FULL PRESCRIBING INFORMATION: CONTENTS*
- SHISEIDO FUTURE SOLUTION LX DAYTIME PROTECTIVE Uses
- Warnings
- Directions
- Inactive Ingredients
- SHISEIDO FUTURE SOLUTION LX DAYTIME PROTECTIVE Other information
- Questions or comments?
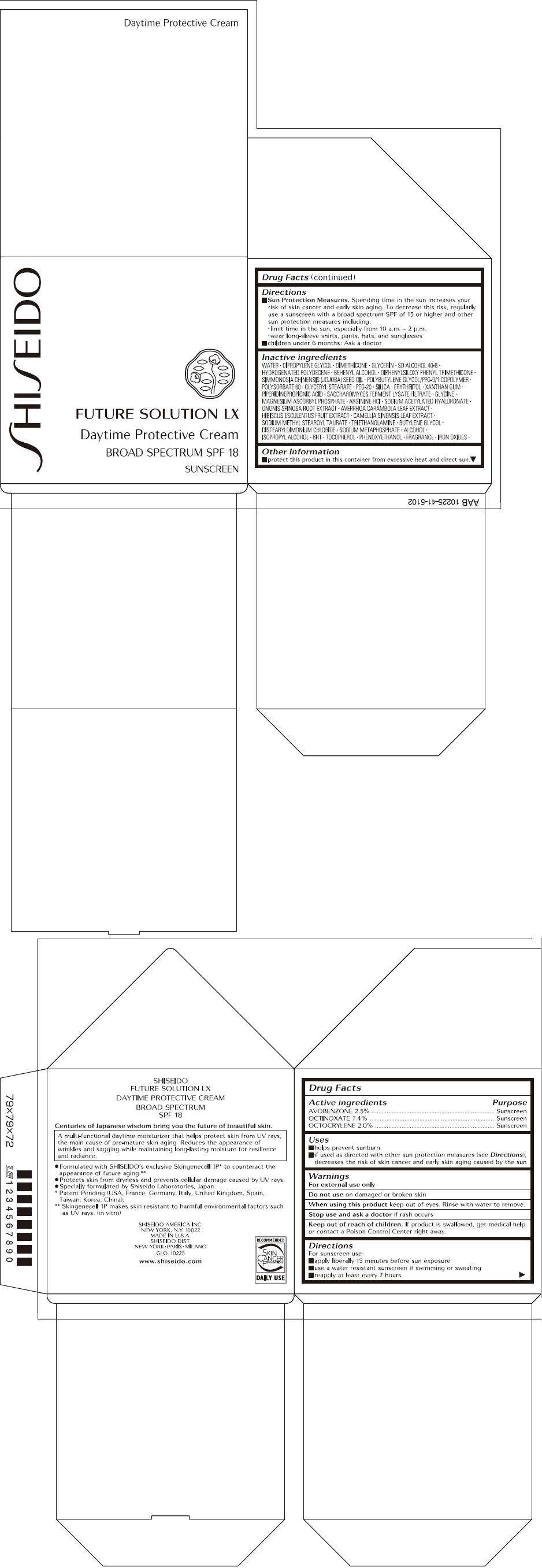
- PRINCIPAL DISPLAY PANEL - 50.2 g Jar Carton
FULL PRESCRIBING INFORMATION
Drug Facts
Active ingredient
Purpose
| ACTIVE INGREDIENTS: | Purpose |
|---|---|
| AVOBENZONE 2.5% | Sunscreen |
| OCTINOXATE 7.4% | Sunscreen |
| OCTOCRYLENE 2.0% | Sunscreen |
SHISEIDO FUTURE SOLUTION LX DAYTIME PROTECTIVE Uses
- helps prevent sunburn
- if used as directed with other sun protection measures (see Directions ), decreases the risk of skin cancer and early skin aging caused by the sun
Warnings
For external use only
Do not use on damaged or broken skin
When using this product keep out of eyes. Rinse with water to remove.
Stop use and ask a doctor if rash occurs
Keep out of reach of children. If product is swallowed, get medical help or contact a Poison Control Center right away.
Directions
For sunscreen use:
- apply liberally 15 minutes before sun exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeve shirts, pants, hats, and sunglasses
- children under 6 months: Ask a doctor
Inactive Ingredients
WATER, DIPROPYLENE GLYCOL, DIMETHICONE, GLYCERIN, SD ALCOHOL 40-B, HYDROGENATED POLYDECENE, BEHENYL ALCOHOL, DIPHENYLSILOXY PHENYL TRIMETHICONE, SIMMONDSIA CHINENSIS (JOJOBA) SEED OIL, POLYBUTYLENE GLYCOL/PPG-9/1 COPOLYMER, POLYSORBATE 60, GLYCERYL STEARATE, PEG-20, SILICA, ERYTHRITOL, XANTHAN GUM, PIPERIDINEPROPIONIC ACID, SACCHAROMYCES FERMENT LYSATE FILTRATE, GLYCINE, MAGNESIUM ASCORBYL PHOSPHATE, ARGININE HCl, SODIUM ACETYLATED HYALURONATE, ONONIS SPINOSA ROOT EXTRACT, AVERRHOA CARAMBOLA LEAF EXTRACT, HIBISCUS ESCULENTUS FRUIT EXTRACT, CAMELLIA SINENSIS LEAF EXTRACT, SODIUM METHYL STEAROYL TAURATE, TRIETHANOLAMINE, BUTYLENE GLYCOL, DISTEARYLDIMONIUM CHLORIDE, SODIUM METAPHOSPHATE, ALCOHOL, ISOPROPYL ALCOHOL, BHT, TOCOPHEROL, PHENOXYETHANOL, FRAGRANCE, IRON OXIDES,
SHISEIDO FUTURE SOLUTION LX DAYTIME PROTECTIVE Other information
- protect this product in this container from excessive heat and direct sun.
Questions or comments?
Call toll free 1-800-906-7503
PRINCIPAL DISPLAY PANEL - 50.2 g Jar Carton
SHISEIDO
FUTURE SOLUTION LX
Daytime Protective Cream
BROAD SPECTRUM SPF 18
SUNSCREEN
SHISEIDO FUTURE SOLUTION LX DAYTIME PROTECTIVEAVOBENZONE, OCTINOXATE, and OCTOCRYLENE CREAM
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||