Shopko Bacitracin Zinc
Shopko Stores Operating Co., LLC
Taro Pharmaceuticals U.S. A., Inc.
ShopkoBacitracin Zinc Ointment USP
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active ingredient (each gram contains)
- Purpose
- Shopko Bacitracin Zinc Uses
- Warnings
- Directions
- Shopko Bacitracin Zinc Other information
- Inactive ingredients
- Questions?
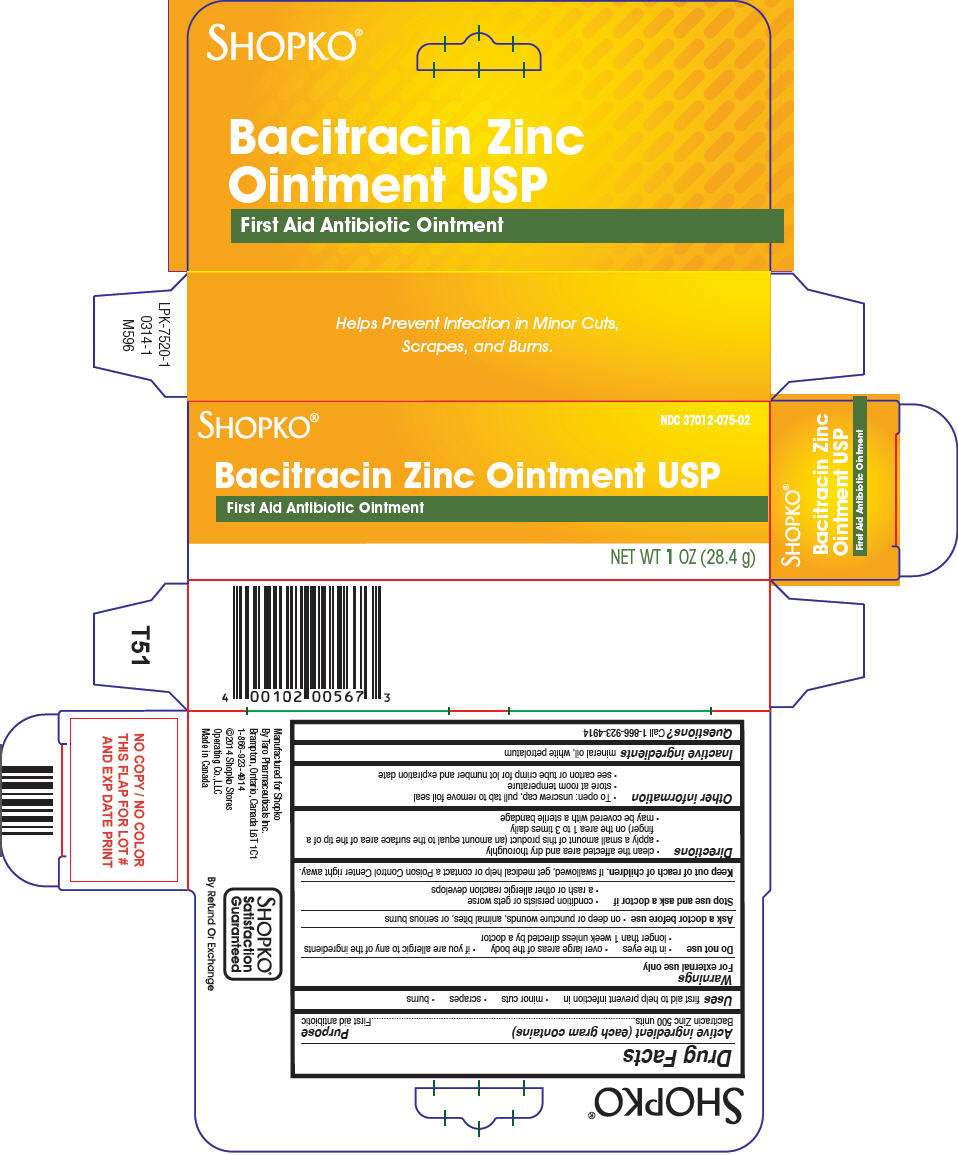
- PRINCIPAL DISPLAY PANEL - 28.4 g Tube Carton
FULL PRESCRIBING INFORMATION
Drug Facts
Active ingredient (each gram contains)
Bacitracin Zinc 500 units
Purpose
First aid antibiotic
Shopko Bacitracin Zinc Uses
first aid to help prevent infection in
- minor cuts
- scrapes
- burns
Warnings
For external use only
Do not use
- in the eyes
- over large areas of the body
- if you are allergic to any of the ingredients
- longer than 1 week unless directed by a doctor
Ask a doctor before use
- on deep or puncture wounds, animal bites, or serious burns
Stop use and ask a doctor if
- condition persists or gets worse
- a rash or other allergic reaction develops
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- clean the affected area and dry thoroughly
- apply a small amount of this product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily
- may be covered with a sterile bandage
Shopko Bacitracin Zinc Other information
- To open: unscrew cap, pull tab to remove foil seal
- store at room temperature
- see carton or tube crimp for lot number and expiration date
Inactive ingredients
mineral oil, white petrolatum
Questions?
Call 1-866-923-4914
PRINCIPAL DISPLAY PANEL - 28.4 g Tube Carton
NDC 37012-075-02
SHOPKO®
Bacitracin Zinc Ointment USP
First Aid Antibiotic Ointment
NET WT 1 OZ (28.4 g)
Shopko Bacitracin ZincBacitracin Zinc OINTMENT
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||