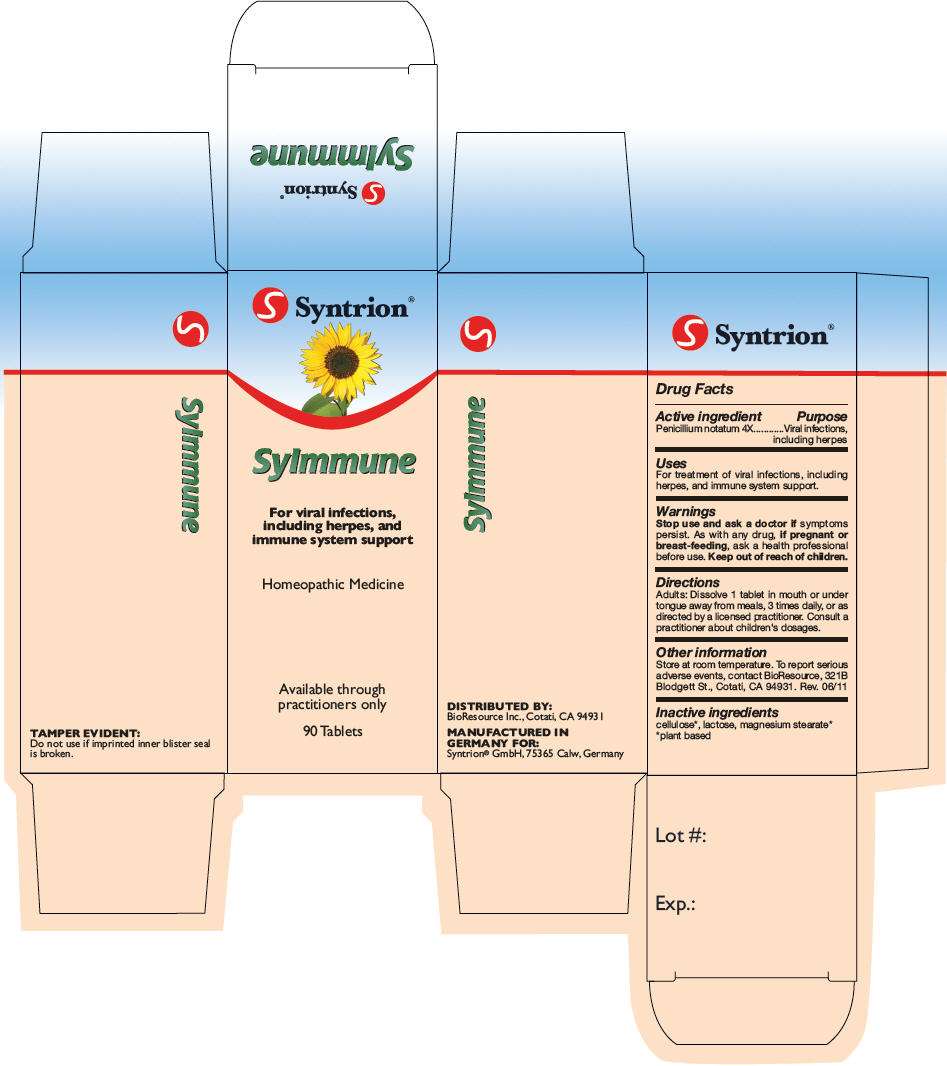
SyImmune
Syntrion SyImmune
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active ingredient
- Purpose
- SyImmune Uses
- Warnings
- Directions
- SyImmune Other information
- Inactive ingredients
- PRINCIPAL DISPLAY PANEL - 90 Tablet Bottle Carton
FULL PRESCRIBING INFORMATION
Drug Facts
Active ingredient
Penicillium notatum 4X
Purpose
Viral infections, including herpes
SyImmune Uses
For treatment of viral infections, including herpes, and immune system support.
Warnings
Stop use and ask a doctor if symptoms persist.
As with any drug, if pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children.
Directions
Adults
Dissolve 1 tablet in mouth or under tongue away from meals, 3 times daily, or as directed by a licensed practitioner. Consult a practitioner about children's dosages.
SyImmune Other information
Store at room temperature. To report serious adverse events, contact BioResource, 321B Blodgett St., Cotati, CA 94931. Rev. 06/11
Inactive ingredients
cellulose

DISTRIBUTED BY:
BioResource Inc., Cotati, CA 94931
MANUFACTURED IN
GERMANY FOR:
Syntrion® GmbH, 75365 Calw, Germany
PRINCIPAL DISPLAY PANEL - 90 Tablet Bottle Carton
S Syntrion ®
SyImmune
For viral infections,
including herpes, and
immune system support
Homeopathic Medicine
Available through
practitioners only
90 Tablets
SyImmunepenicillium chrysogenum var. chrysogenum TABLET
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||