symmetry Non-Alcohol Foaming Hand Sanitizer with Allantoin and Aloe
symmetry Non-Alcohol Foaming Hand Sanitizer with Allantoin & Aloe
FULL PRESCRIBING INFORMATION: CONTENTS*
- symmetry Non-Alcohol Foaming Hand Sanitizer with Allantoin & Aloe
- Active ingredient
- symmetry Non-Alcohol Foaming Hand Sanitizer with Allantoin and Aloe Uses
- Warnings
- Directions
- Inactive ingredients
- Questions?
- Representative Labeling
FULL PRESCRIBING INFORMATION
symmetry Non-Alcohol Foaming Hand Sanitizer with Allantoin & Aloe
Active ingredient
Benzalkonium Chloride 0.12%
Purpose
Antibacterial
symmetry Non-Alcohol Foaming Hand Sanitizer with Allantoin and Aloe Uses
- Hand sanitizer to help reduce bacteria on the skin could cause disease
- Recommended for repeated use
Warnings
For external use only.
When using this product
do not use in or near eyes. If in eyes, flush thoroughly with water.
If irritation or rash
appears and persists, stop use and see physician.
Keep out of reach of children.
If swallowed, call a physician or Poison Control Center immediately.
Directions
- Dispense an adequate amount of hand sanitizer
- Rub hands together until completely dry
Inactive ingredients
Water (Aqua), Cocamidopropyl Betaine, Lauramine Oxide, Tetrasodium EDTA, Allantoin, Fragrance (Parfum), Aloe Barbadensis Leaf Juice, Methylchloroisothiazolinone, Methylisothiazolinone
Questions?
Call: Buckeye International, Inc. - 314-291-1900 Monday through Friday - 8:00 a.m. to 5:00 p.m. CST
Representative Labeling
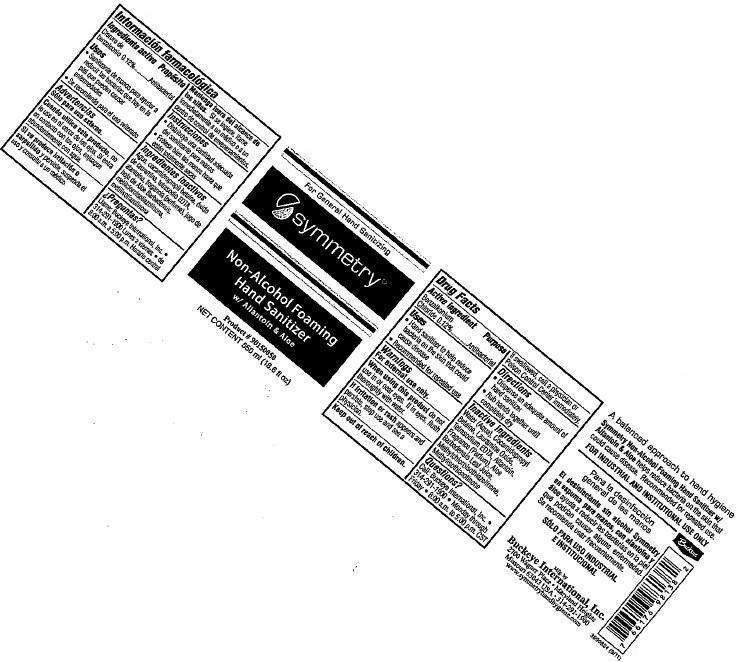
symmetry Non-Alcohol Foaming Hand Sanitizer with Allantoin and AloeBENZALKONIUM CHLORIDE LIQUID
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!