Tamsulosin Hydrochloride
FULL PRESCRIBING INFORMATION: CONTENTS*
- TAMSULOSIN HYDROCHLORIDE DESCRIPTION
- CLINICAL PHARMACOLOGY
- PHARMACOKINETICS
- CLINICAL STUDIES
- TAMSULOSIN HYDROCHLORIDE CONTRAINDICATIONS
- INDICATIONS & USAGE
- WARNINGS
- PRECAUTIONS
- INFORMATION FOR PATIENTS
- LABORATORY TESTS
- PREGNANCY
- GERIATRIC USE
- NURSING MOTHERS
- PEDIATRIC USE
- CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
- TAMSULOSIN HYDROCHLORIDE ADVERSE REACTIONS
- OVERDOSAGE
- DOSAGE & ADMINISTRATION
- HOW SUPPLIED
- INFORMATION FOR PATIENTS
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
FULL PRESCRIBING INFORMATION
TAMSULOSIN HYDROCHLORIDE DESCRIPTION
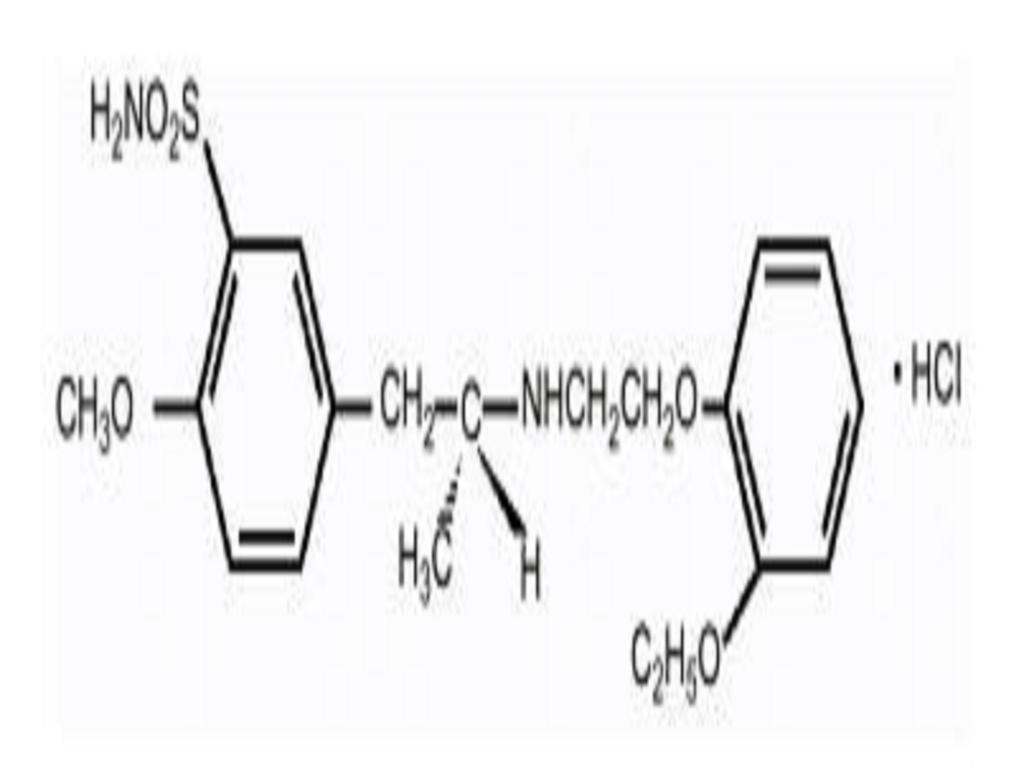
CLINICAL PHARMACOLOGY
PHARMACOKINETICS
Absorption
Effect of Food
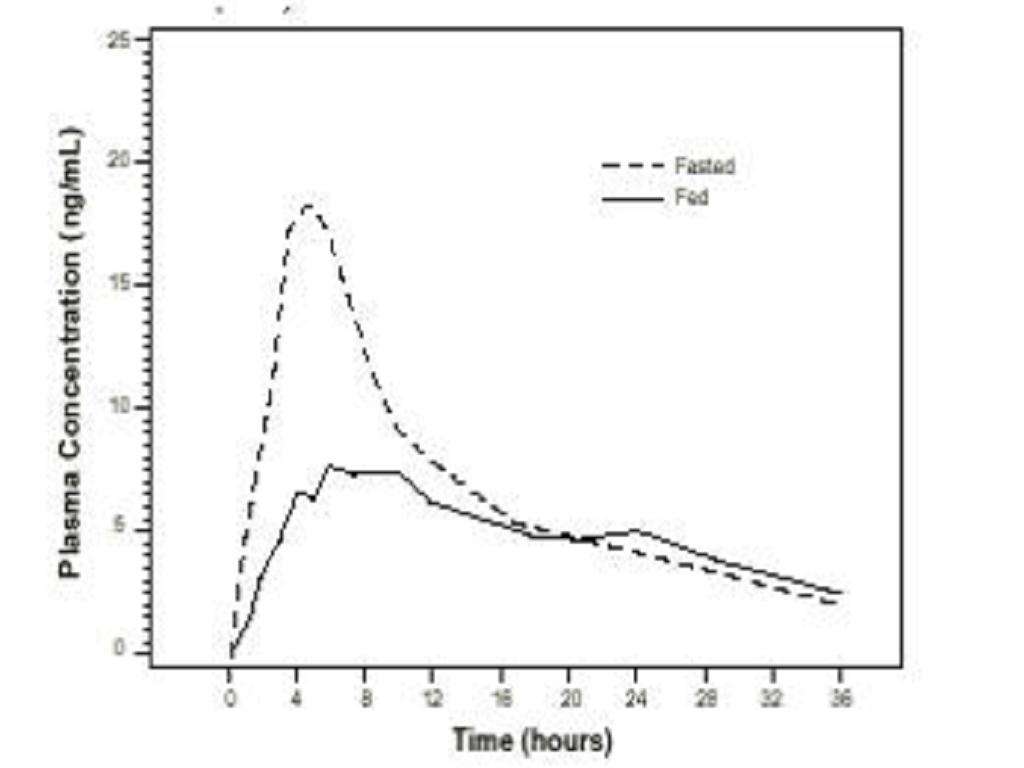
Pharmacokinetic ParameterLight BreakfastFastedLight BreakfastHigh-Fat BreakfastFasted
Distribution
Metabolism
Drug-Drug InteractionsCytochrome P450 Inhibition
Excretion
Special Populations
Geriatrics (Age)
Renal Dysfunction
Hepatic Dysfunction
Drug-Drug Interactions
Nifedipine, Atenolol, Enalapril
Warfarin
Digoxin and Theophylline
Furosemide
The pharmacokinetic and pharmacodynamic interaction between Flomax(tamsulosin hydrochloride) capsules 0.8 mg/day (steady-state) and furosemide 20 mg intravenously (single dose) was evaluated in ten healthy volunteers (age range 21-40 years). FLOMAX capsules had no effect on the pharmacodynamics (excretion of electrolytes) of furosemide. While furosemide produced an 11% to 12% reduction in tamsulosin hydrochloride Cmax and AUC, these changes are expected to be clinically insignificant and do not require adjustment of the FLOMAX capsules dosage.
Cytochrome P450 Inhibition:
Cimetidine
The effects of cimetidine at the highest recommended dose (400 mg every six hours for six days) on the pharmacokinetics of a single FLOMAX capsule 0.4 mg dose was investigated in ten healthy volunteers (age range 21-38 years). Treatment with cimetidine resulted in a significant decrease (26%) in the clearance of tamsulosin hydrochloride, which resulted in a moderate increase in tamsulosin hydrochloride AUC (44%). Therefore, FLOMAX capsules should be used with caution in combination with cimetidine, particularly at doses higher than 0.4 mg.
Strong Inhibitors of CYP3A4 or CYP2D6
The effects of ketoconazole (a strong inhibitor of CYP3A4) at 400 mg every day on the pharmacokinetics of a single FLOMAX capsule 0.4 mg dose was investigated in 24 healthy volunteers (age range from 23 to 47 years). Treatment with ketoconazole resulted in a Cmax and AUC that increased by a factor of 2.2 and 2.8, respectively. Therefore, FLOMAX 0.4 mg capsules should be used with caution in combination with strong inhibitors of CYP3A4. Doses above 0.4 mg should not be used in combination with strong inhibitors of CYP3A4 (e.g., ketoconazole).
The effects of paroxetine (a strong inhibitor of CYP2D6) at 20 mg every day on the pharmacokinetics of a single FLOMAX capsule 0.4 mg dose was investigated in 23 healthy volunteers (age range from 23 to 48 years). Treatment with paroxetine resulted in a Cmax and AUC that increased by a factor of 1.3 and 1.6, respectively. Therefore, FLOMAX capsules should be used with caution in combination with strong inhibitors of CYP2D6, particularly at doses higher than 0.4 mg (e.g., 0.8 mg).
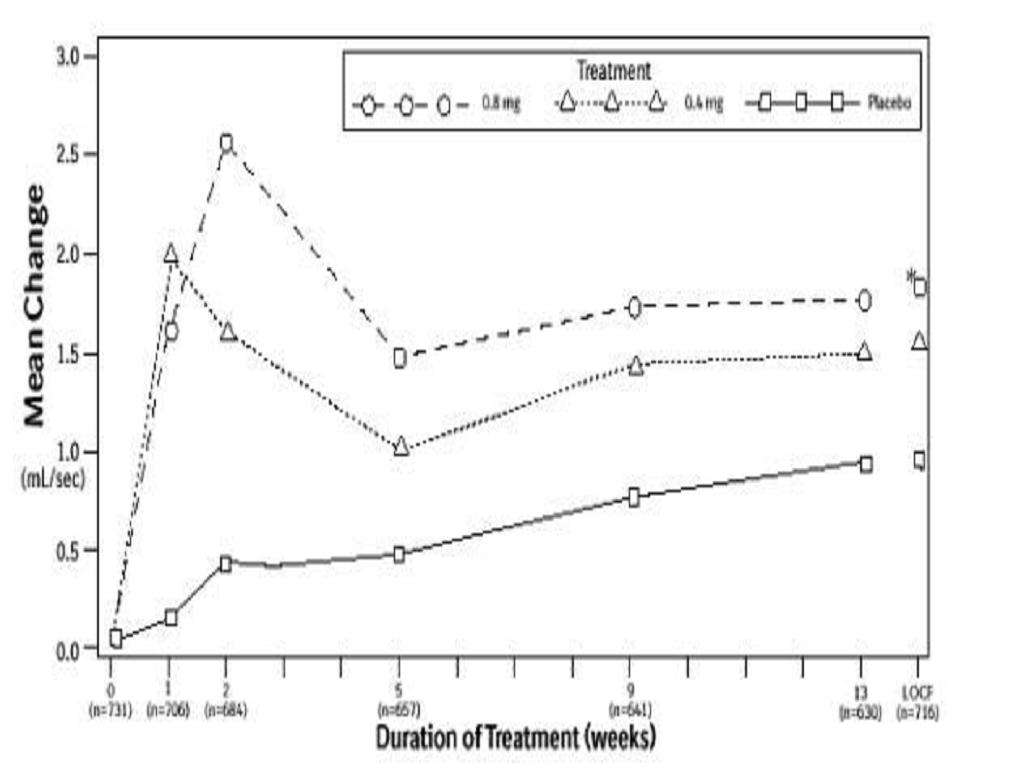
CLINICAL STUDIES
Total AUA Symptom ScorePeak Urine Flow RateMean Baseline ValueMean ChangeMean Baseline ValueMean ChangeStudy 1Study 2

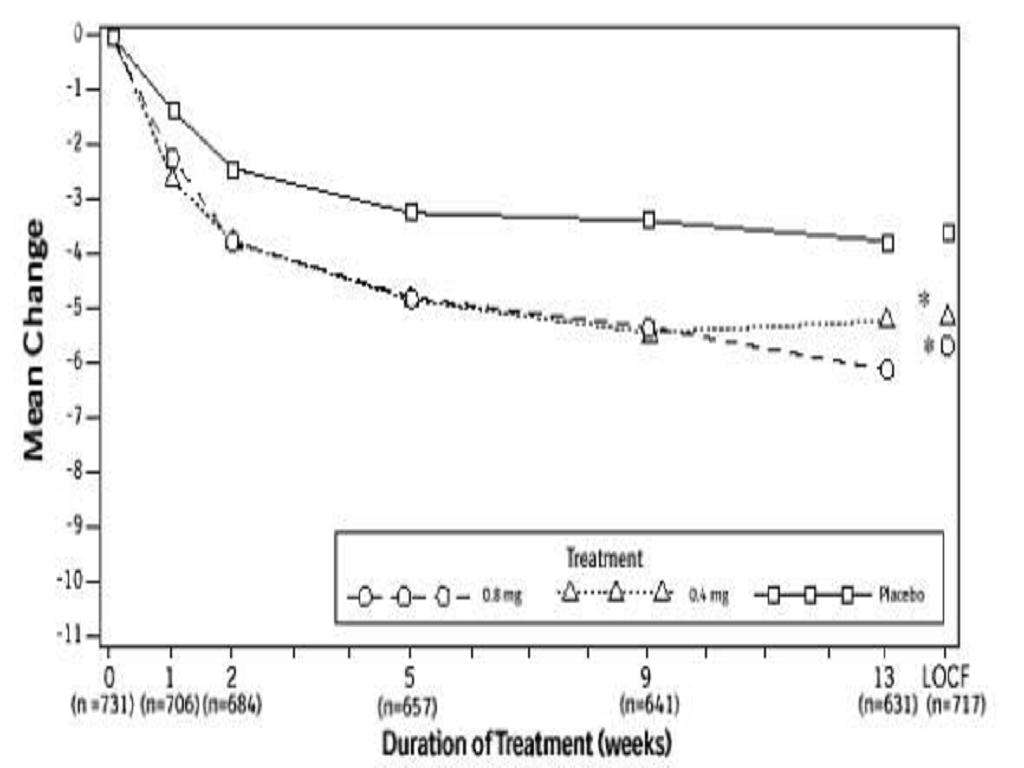
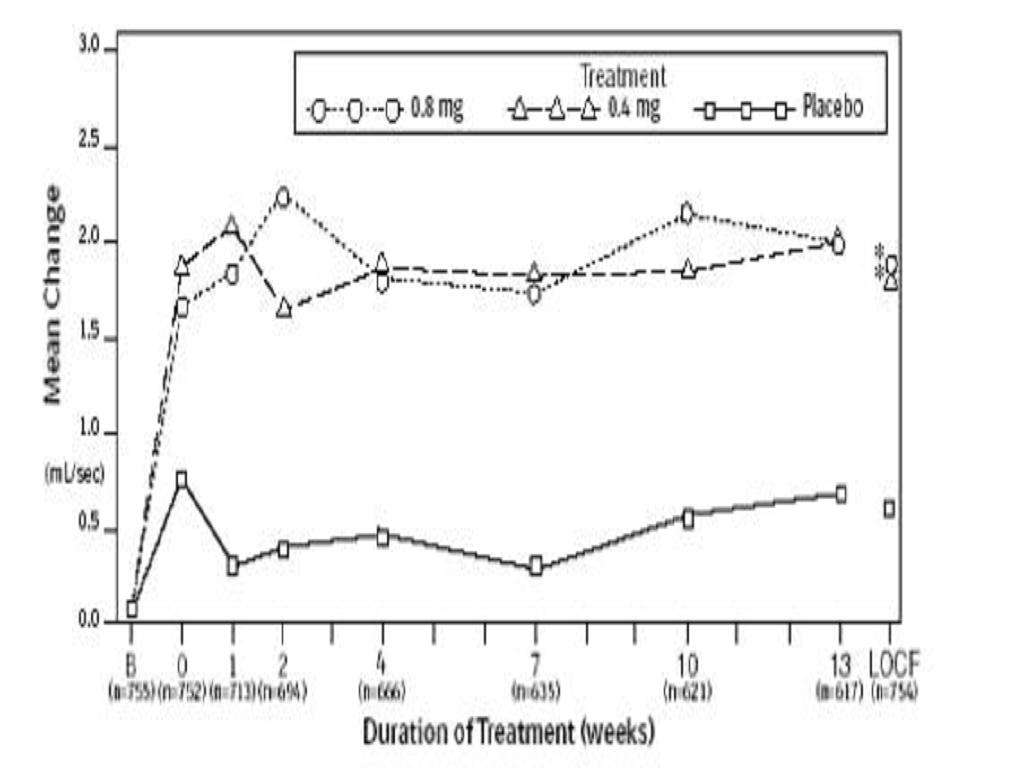
TAMSULOSIN HYDROCHLORIDE CONTRAINDICATIONS
INDICATIONS & USAGE
WARNINGS
ADVERSE REACTIONSPRECAUTIONS, Information for Patients
PRECAUTIONS
GeneralCLINICAL PHARMACOLOGYDrug-Drug Interactions
WARNINGS
Drug-Drug Interactions
INFORMATION FOR PATIENTS
LABORATORY TESTS
PREGNANCY
Teratogenic EffectsGERIATRIC USE
CLINICAL PHARMACOLOGY, PharmacokineticsSpecial PopulationsGeriatrics (Age))NURSING MOTHERS
PEDIATRIC USE
CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
TAMSULOSIN HYDROCHLORIDE ADVERSE REACTIONS
BODY SYSTEM/ ADVERSE EVENTFLOMAX CAPSULES GROUPSPLACEBO0.4 mg n=5020.8 mg n=492n=493BODY AS WHOLENERVOUS SYSTEMRESPIRATORY SYSTEMDIGESTIVE SYSTEMUROGENITAL SYSTEMSPECIAL SENSES
WARNINGS
Post-Marketing Experience
PRECAUTIONS, General
OVERDOSAGE
WARNINGSADVERSE REACTIONSDOSAGE & ADMINISTRATION
HOW SUPPLIED
INFORMATION FOR PATIENTS
Tamsulosin Hydrochloride Capsules USP*, 0.4 mgWhat is tamsulosin hydrochloride?
-
● Tamsulosin hydrochloride capsules are not for women.
-
● Tamsulosin hydrochloride capsules are not for children.
What should I tell my doctor before using tamsulosin hydrochloride capsules, USP?
Before taking tamsulosin hydrochloride capsules, USP tell your doctor about all your medical conditions including:
-
● any kidney or liver problems.
-
● any history of low blood pressure.
-
● any allergies to sulfa or any other medicines.
-
● if you are planning to have cataract surgery.
-
● any prescription medicines, including blood pressure medicines.
-
● any non-prescription medicines, including vitamins and herbal supplements.
How should I take tamsulosin hydrochloride capsules, USP?
-
● Take tamsulosin hydrochloride capsules exactly as prescribed by your doctor.
-
● Do not crush, chew, or open tamsulosin hydrochloride capsules.
-
● Take tamsulosin hydrochloride capsules one time each day, about 30 minutes after the same meal each day. For example, you may take tamsulosin hydrochloride capsules 30 minutes after dinner each day.
-
● If you miss a dose of tamsulosin hydrochloride capsules, take it as soon as you remember. If you miss your dose for the whole day, continue with your next dose on your regular schedule. Do not take two doses at the same time.
-
● If you stop or forget to take tamsulosin hydrochloride capsules for several days, talk with your doctor before starting again.
-
● If you take more tamsulosin hydrochloride capsules than prescribed, call your doctor right away.
-
● Decreased blood pressure when changing positions.Tamsulosin hydrochloride capsules may cause a sudden drop in blood pressure upon standing, especially after the first dose or when changing doses.
-
● fainting
-
● dizziness
-
● lightheadedness
-
● Allergic reactions.Make your doctor aware of any allergic reactions you may experience while taking tamsulosin hydrochloride.
-
● rash
-
● itching
-
● hives
-
● swelling of face, tongue, or throat
-
● difficulty breathing
-
● A painful erection that will not go away.Tamsulosin hydrochloride capsules can cause a painful erection (priapism), which cannot be relieved by having sex. If this happens, get medical help right away. If priapism is not treated, you may not be able to get an erection in the future.
-
● Eye problems during cataract surgery.During cataract surgery, a condition called intraoperative floppy iris syndrome (IFIS) can happen if you take or have taken tamsulosin hydrochloride capsules. If you need to have cataract surgery, be sure to tell your surgeon if you take or have taken tamsulosin hydrochloride capsules.
-
● runny nose
-
● dizziness
-
● decreased semen
What should I avoid while taking tamsulosin hydrochloride capsules, USP?
How do I store tamsulosin hydrochloride capsules, USP?
Keep tamsulosin hydrochloride capsules, USP and all medicines out of the reach of children.
General information
What are the ingredients in tamsulosin hydrochloride capsules, USP?
-
● Active Ingredient: tamsulosin hydrochloride
-
● Inactive Ingredients: microcrystalline cellulose, magnesium stearate, methacrylic acid copolymer dispersion, talc, triethyl citrate, FD and C blue # 1, FD and C red # 40, FD and C yellow # 10, titanium dioxide, gelatin, shellac, dehydrated alcohol, isopropyl alcohol, butyl alcohol, propylene glycol, strong ammonia solution, black iron oxide and potassium hydroxide
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION

PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Tamsulosin HydrochlorideTamsulosin Hydrochloride CAPSULE
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!