Thyreoidea compositum
Thyreoidea compositum oral vial
FULL PRESCRIBING INFORMATION: CONTENTS*
- THYREOIDEA COMPOSITUM DESCRIPTION
- INDICATION AND USAGE
- THYREOIDEA COMPOSITUM DOSAGE AND ADMINISTRATION
- THYREOIDEA COMPOSITUM CONTRAINDICATIONS
- WARNINGS AND PRECAUTIONS
- THYREOIDEA COMPOSITUM ADVERSE REACTIONS
- OVERDOSAGE
- CLINICAL PHARMACOLOGY
- DOSAGE
FULL PRESCRIBING INFORMATION
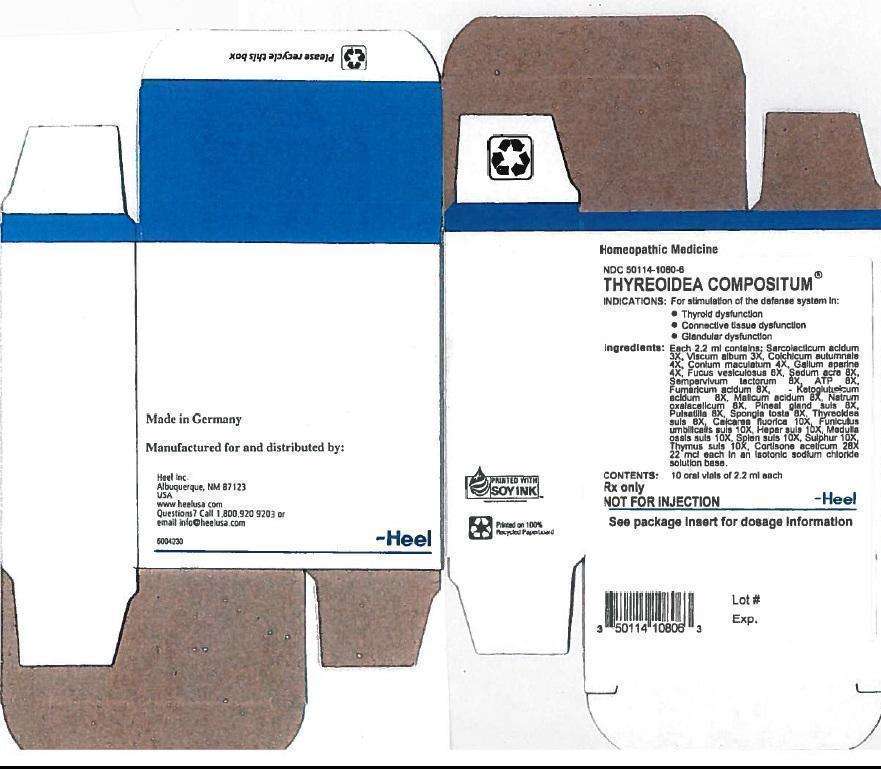
THYREOIDEA COMPOSITUM DESCRIPTION
Each 2.2 ml ampule contains:
|
Ingredient Name |
Potency |
Quantity |
|
Sarcolactium acidum |
3x |
22µ |
|
Viscum album |
3x |
22µ |
|
ATP |
8x |
22µ |
|
alpha-Ketoglutaricum acidum |
8x |
22µ |
|
Calcarea fluorica |
10x |
22µ |
|
Thyroidinum |
8x |
22µ |
|
Galium aparine |
4x |
22µ |
|
Cortisone aceticum |
28x |
22µ |
|
Fumaricum acidum |
8x |
22µ |
|
Sempervivum tectorum |
6x |
22µ |
|
Malicum acidum |
8x |
22µ |
|
Colchicum autumnale |
4x |
22µ |
|
Sedum acre |
6x |
22µ |
|
Conium maculatum |
4x |
22µ |
|
Hepar suis |
10x |
22µ |
|
Medulla ossis suis |
10x |
22µ |
|
Splen suis |
10x |
22µ |
|
Thymus suis |
10x |
22µ |
|
Thyreoidea suis |
8x |
22µ |
|
Funiculus umbilicalis suis |
10x |
22µ |
|
Spongia tosta |
8x |
22µ |
|
Fucus vesiculosus |
6x |
22µ |
|
Natrum oxalceticum |
8x |
22µ |
|
Sulphur |
10x |
22µ |
|
Pulsatilla |
8x |
22µ |
Inactive Ingredient: Isotonic Sodium Chloride solution
INDICATION AND USAGE
Thyreoidea compositum® Oral Vials is a homeopathic drug product indicated for the stimulation of the defense system in thyroid dysfunction, connective tissue dysfunction, and glandular dysfunction
THYREOIDEA COMPOSITUM DOSAGE AND ADMINISTRATION
Dosage:
Adults and children above 6 years: 1 vial orally 1-3 times daily
Children up to 6 years: ½ vial orally 1-3 times daily
THYREOIDEA COMPOSITUM CONTRAINDICATIONS
Thyreoidea compositum® Oral Vials are contraindicated in patients with known hypersensitivity to Thyreoidea compositum® or any of its ingredients.
WARNINGS AND PRECAUTIONS
Warnings and Precautions
None
THYREOIDEA COMPOSITUM ADVERSE REACTIONS
No adverse events have been reported with a causal relationship Thyreoidea compositum® Oral Vials.
OVERDOSAGE
Overdosage: No negative effects of an overdose have been reported and none are expected due to the homeopathic dilutions
CLINICAL PHARMACOLOGY
Mechanism of Action
The exact mechanism of Thyreoidea compositum® Oral Vials is not fully understood.
Pharmacodynamics
Not applicable for homeopathic medicinal products.
DOSAGE
1 oral vial containing 2.2ml solution for oral administration
Thyreoidea compositumLACTIC ACID, L-,VISCUM ALBUM FRUITING TOP,COLCHICUM AUTUMNALE BULB,CONIUM MACULATUM FLOWERING TOP,GALIUM APARINE,FUCUS VESICULOSUS, SEDUM ACRE,SEMPERVIVUM TECTORUM LEAF, ADENOSINE CYCLIC PHOSPHATE, FUMARIC ACID, ALPHA.-KETOGLUTARIC ACID, MALIC ACID, SODIUM DIETHYL OXALACETATE ,SUS SCROFA PINEAL GLAND ,PULSATILLA VULGARIS, SPONGIA OFFICINALIS SKELETON, ROASTED,THYROID, PORCINE,CALCIUM FLUORIDE, SUS SCROFA UMBILICAL CORD,PORK LIVER ,SUS SCROFA BONE MARROW, SUS SCROFA SPLEEN,SUS SCROFA THYMUS, CORTISONE SOLUTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||