Tired Eye Relief
Tired Eye Relief
FULL PRESCRIBING INFORMATION
Drug Facts
Conium maculatum 6X
burning, redness
Natrium muriaticum 6X
burning, redness, eyestrain
Ruta graveolens 6X
burning, eye strain, tired sensation
Senega officinalis 6X
redness, dryness tired sensation
According to homeopathic principles, the active ingredients in this medication temporarily relieve minor symptoms of eye fatigue such as:
- stressed, tired eyes
- burning & dryness due to eye fatigue
- overstrained eyes (stress, PC, TV, reading, driving, jetlag)
- redness
- For external use only.
- Initial exacerbation of symptoms may occur.
- Replace cap tightly after every use.
- To avoid contamination, do not touch the tip of the container to any surface.
- Use within 30 days of opening. Expiration date only refers to unopened bottle.
- Contact wearers: consult a physician prior to using.
- if the solution changes color or becomes cloudy
- symptoms worsen or persist for more than 72 hours
- changes in vision occur
- you experience eye pain
ask a health professional before use.
If swallowed, get medical help or contact a Poison Control Center right away.
For adults and children age
2 and over:
- remove tamper-evident seal from neck of bottle
- twist cap off bottle
- squeeze plastic tip to release 2-3 drops into eye
- apply as needed
- replace cap after use
Active ingredients are manufactured according to homeopathic principles.
Borate buffer, Purified water, Silver sulphate (as preservative), Sodium nitrate
Reach our representatives at 1-800-240-9780 or getinfo@similasanusa.com.
www.SimilasanUSA.com
NEW
NDC
59262-357-11
Similasan
Tired Eye Relief
STERILE EYE DROPS
10
mL
/0.33 fl oz
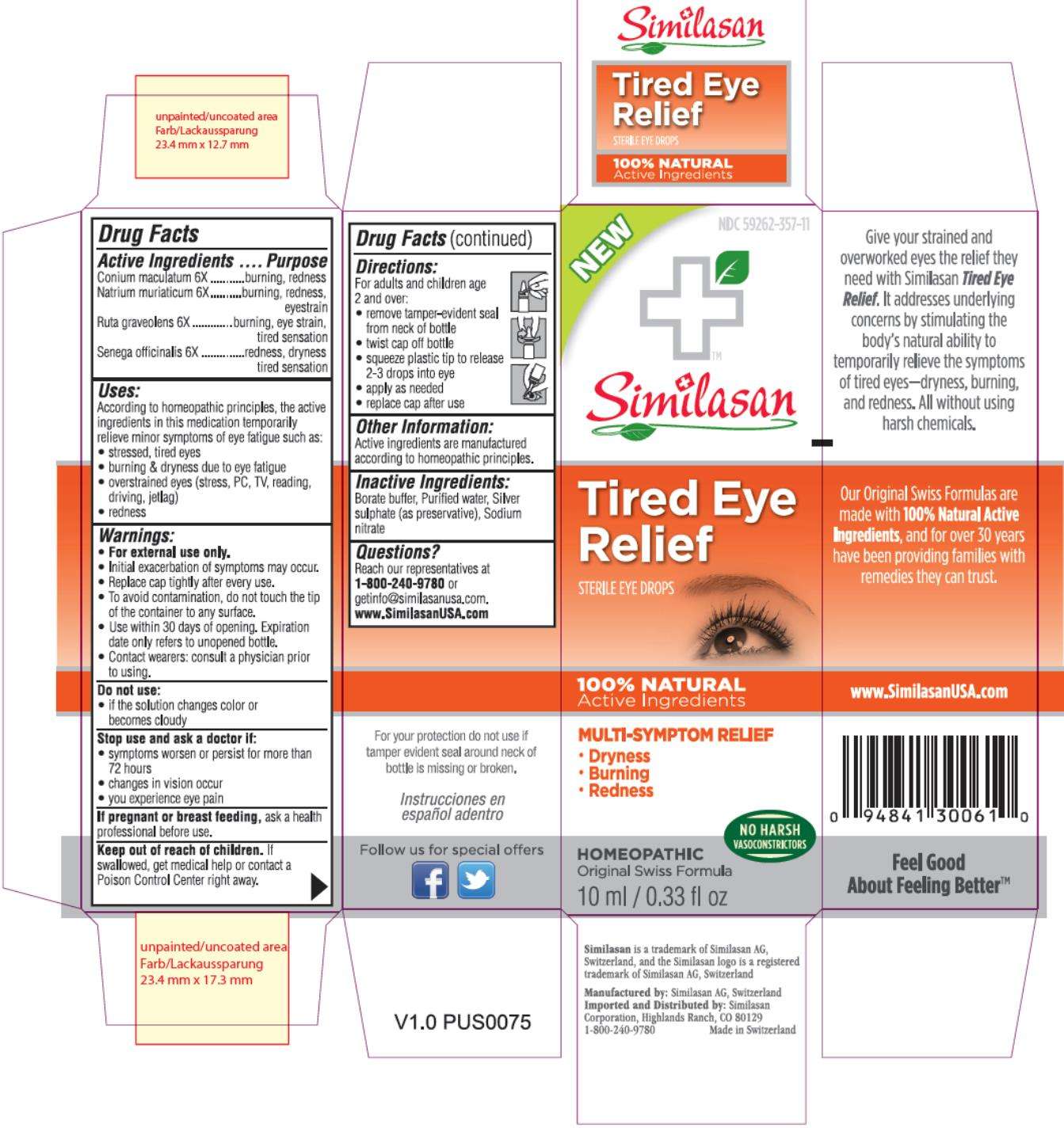
Tired Eye Reliefconium maculatum flowering top, sodium chloride, ruta graveolens flowering top and polygala senega root SOLUTION/ DROPS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||