Triaminic
Novartis Consumer Health, Inc.
Drug Facts
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active ingredient
- Purpose
- Triaminic Uses
- Warnings
- Do not use
- Ask Doctor before use if your child has
- When using this product
- Stop use and ask a doctor if
- Keep Out of Reach of Children
- Directions
- Triaminic Other information
- Inactive ingredients
- Questions or comments?
- Additional Information Listed on Other Panels
- Principal Display - Tropical
FULL PRESCRIBING INFORMATION
Active ingredient
Guaifenesin 50 mg
Phenylephrine HCl 2.5 mg
Purpose
Expectorant
Nasal decongestant
Triaminic Uses
- helps loosen phlegm (mucus) and thins bronchial secretions to make coughs more productive
- temporarily relieves nasal and sinus congestion as may occur with a cold
Warnings
Do not use
- in a child under 4 years of age
- in a child who is taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease), or for 2 weeks after stopping the MAOI drug. If you do not know if the child’s prescription drug contains an MAOI, ask a doctor or pharmacist before giving this product.
Ask Doctor before use if your child has
- heart disease • high blood pressure
- thyroid disease • diabetes
- cough that occurs with too much phlegm (mucus)
- chronic cough that lasts, or as occurs with asthma
When using this product
- do not exceed recommended dosage
Stop use and ask a doctor if
- nervousness, dizziness or sleeplessness occurs
- symptoms do not improve within 7 days or occur with a fever
- cough persists for more than 7 days, comes back or occurs with a fever, rash or persistent headache. These could be signs of a serious condition.
Keep Out of Reach of Children
In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- may be given every 4 hours. Do not give more than 6 doses in 24 hours unless directed by a doctor
-
Age Dose children under 4 years of age do not use children 4 to under 6 years of age 1 teaspoonful (5 mL) children 6 to under 12 years of age 2 teaspoonfuls (10 mL)
Triaminic Other information
- each teaspoonful contains: sodium 3 mg
- store at controlled room temperature 20-25°C (68-77°F)
Inactive ingredients
acesulfame K, benzoic acid, citric acid, D&C yellow #10, edetate disodium, FD&C yellow #6, flavors, maltitol solution, propylene glycol, purified water, sodium citrate
Questions or comments?
call 1-800-452-0051 24 hours a day, 7 days a week
Additional Information Listed on Other Panels
See New Directions and Warnings Information
Children’s
Triaminic®
Syrup
Chest & Nasal Congestion
Guaifenesin (Expectorant)
Phenylephrine HCl (Nasal Decongestant)
- Chest Congestion
- Stuffy Nose
Child-Resistant Safety Cap
TAMPER-EVIDENT FEATURE:
DO NOT USE IF SAFETY SEAL IMPRINTED WITH “TRIAMINIC” AROUND BOTTLE CAP IS BROKEN OR MISSING.
Triaminic® Has More Ways to Help Kids Feel Better!
Great Tasting Liquids
- Free Dosage Cup (enclosed)
- Alcohol Free
Fast-Dissolving Softchews®
- Great Taste
Convenient Thin Strips®
- Melts in an Instant
- Easy to Give/Easy to Take
- No Mess Dosing
Triaminic® provides a broad range of products, in forms created with the needs of children in mind.
Trusted by Parents for over 50 years.
What You Need to Make Your Child Feel Better!
For more information plus helpful tips visit www.triaminic.com or call The Triaminic Clinic®
1-800-KIDS-987 24 hours a day, 7 days a week.
Distr. by: Novartis Consumer Health, Inc.
Parsippany, NJ 07054-0622 ©20xx
Principal Display - Tropical
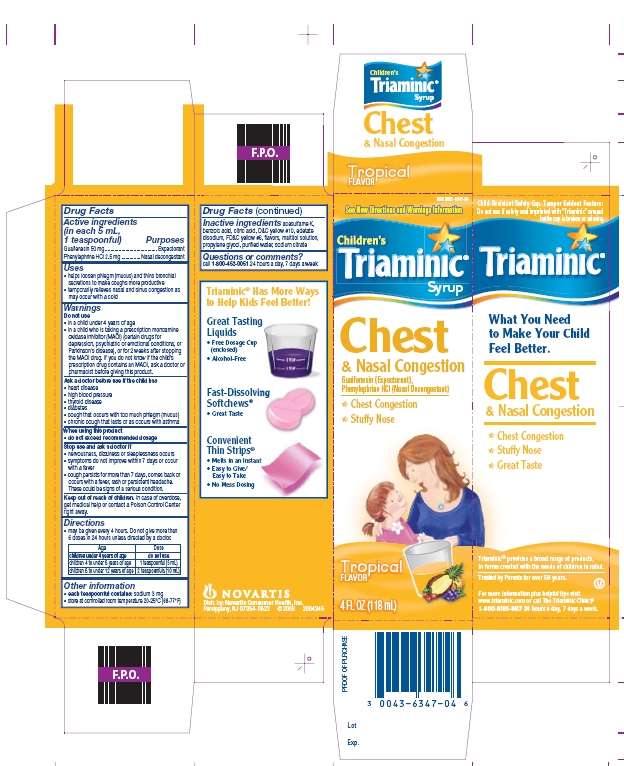
TriaminicGuaifenesin, Phenylephrine HCl SYRUP
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||