Tussin Sugar Free
Premier Value
Aaron Industries, Inc.
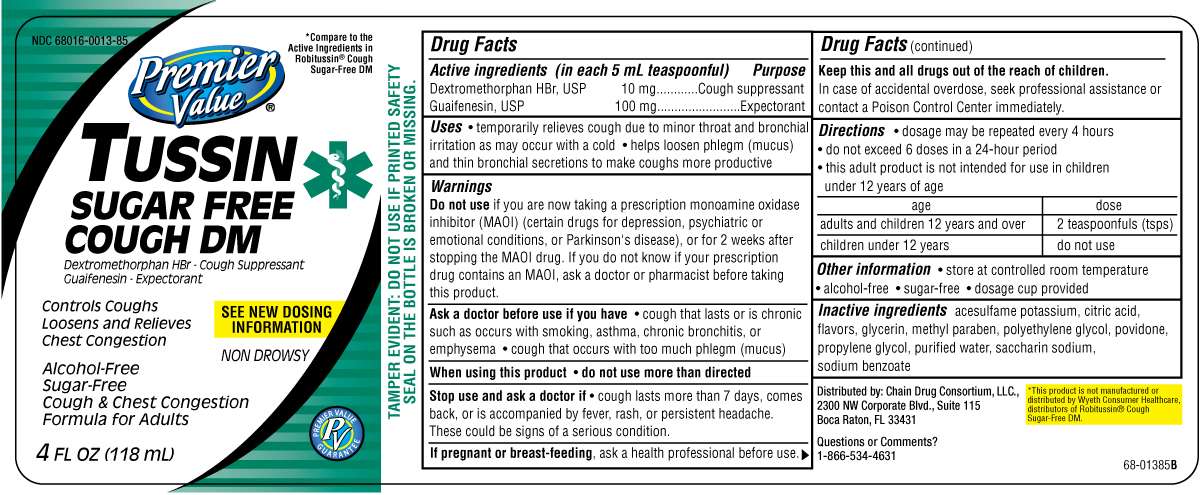
Drug Facts
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active Ingredients
- Purpose
- Tussin Sugar Free Uses
- Warnings
- Ask a Doctor before use if you have
- When using this product
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep this and all drugs out of the reach of children.
- Directions
- Other Information
- Inactive Ingredients
- Principal Display Panel
FULL PRESCRIBING INFORMATION
Active Ingredients
Dextromethorphan HBr,USP 10 mg
Guaifenesin, USP 100 mg
Purpose
Dextromethorphan HBr...................Cough suppressant
Guaifenesin, USP..............................Expectorant
Tussin Sugar Free Uses
• temporarily relieves cough due to minor throat
and bronchial irritation as may occur with a cold
• helps loosen phlegm (mucus) and thin bronchial
secretions to make coughs more productive
Warnings
Do not use if you are now taking a prescription
monoamine oxidase inhibitor (MAOI) (certain
drugs for depression, psychiatric or emotional
conditions, or Parkinson's disease), or for 2 weeks
after stopping the MAOI drug. If you do not know
if your prescription drug contains an MAOI, ask
a doctor or pharmacist before taking this product.
Ask a Doctor before use if you have
• cough that occurs with too much phlegm (mucus)
• cough that lasts or is chronic such as occurs
with smoking, asthma, chronic bronchitis, or
emphysema
When using this product
• do not use more than directed
Stop use and ask a doctor if
• cough lasts more than 7 days, comes back, or is
accompanied by fever, rash, or persistent headache.
These could be signs of a serious condition.
If pregnant or breast-feeding,
ask a health
professional before use.
Keep this and all drugs out of the reach of children.
In case of accidental overdose, seek professional
assistance or contact a Poison Control Center
immediately.
Directions
• do not exceed 6 doses in a 24-hour period
• this adult product is not intended for use in
children under 12 years of age
Age Dose
adults and children 12 years and over 2 teaspoonfuls (tsps) every 4 hours
children under 12 years do not use
Other Information
• store at controlled room temperature
alcohol-free
Inactive Ingredients
acesulfame potassium, citric acid, flavors,
glycerin, methyl paraben, polyethylene glycol,
povidone, propylene glycol, purified water,
saccharin sodium, sodium benzoate
Principal Display Panel
Tussin Sugar FreeDextromethorphan Hydrobromide, GUAIFENESIN LIQUID
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||