Walgreens Corn Remover
Walgreens Corn Remover Strips
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active Ingredient
- Purpose
- Walgreens Corn Remover Uses
- Warnings
- Directions
- Other Information
- Inactive Ingredients
- Questions or Comments?
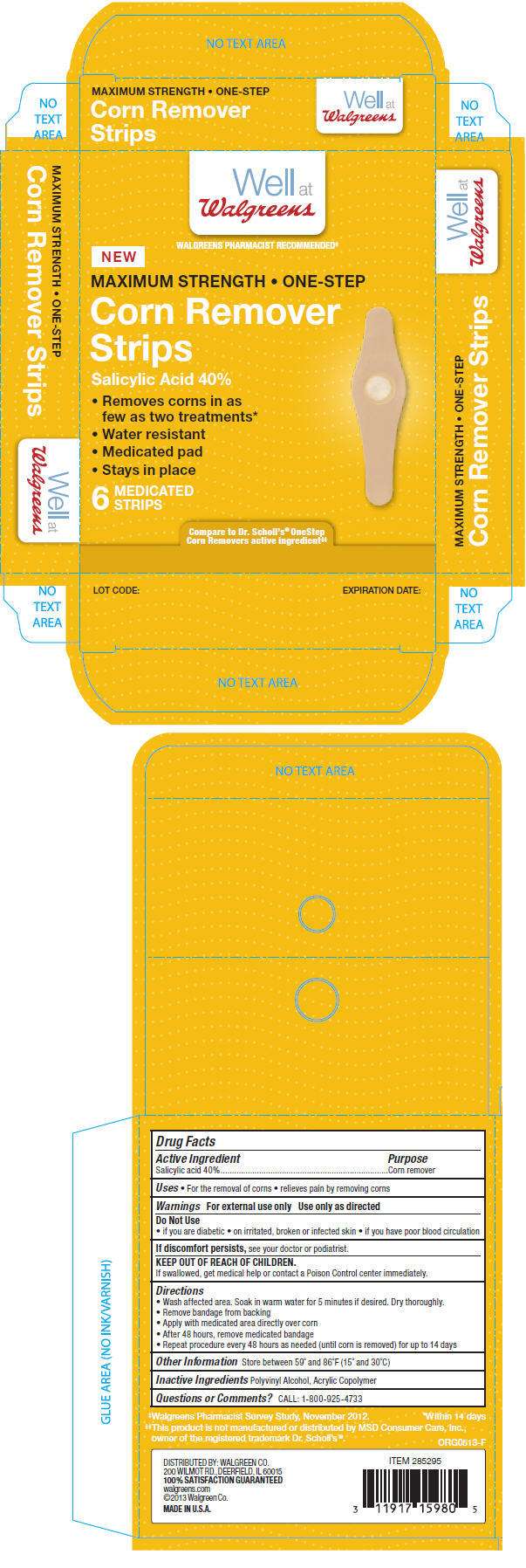
- PRINCIPAL DISPLAY PANEL - 6 Strip Box
FULL PRESCRIBING INFORMATION
Drug Facts
Active Ingredient
Salicylic acid 40%
Purpose
Corn remover
Walgreens Corn Remover Uses
- For the removal of corns
- relieves pain by removing corns
Warnings
For external use only
Use only as directed
Do Not Use
- if you are diabetic
- on irritated, broken or infected skin
- if you have poor blood circulation
If discomfort persists, see your doctor or podiatrist.
KEEP OUT OF REACH OF CHILDREN.
If swallowed, get medical help or contact a Poison Control center immediately.
Directions
- Wash affected area. Soak in warm water for 5 minutes if desired. Dry thoroughly.
- Remove bandage from backing
- Apply with medicated area directly over corn
- After 48 hours, remove medicated bandage
- Repeat procedure every 48 hours as needed (until corn is removed) for up to 14 days
Other Information
Store between 59° and 86°F (15° and 30°C)
Inactive Ingredients
Polyvinyl Alcohol, Acrylic Copolymer
Questions or Comments?
CALL: 1-800-925-4733
DISTRIBUTED BY: WALGREEN CO.
200 WILMOT RD., DEERFIELD, IL 60015
PRINCIPAL DISPLAY PANEL - 6 Strip Box
Well at
Walgreens
WALGREENS PHARMACIST RECOMMENDED‡
NEW
MAXIMUM STRENGTH • ONE-STEP
Corn Remover
Strips
Salicylic Acid 40%
-
Removes corns in as
few as two treatments* - Water resistant
- Medicated pad
- Stays in place
6
MEDICATED
STRIPS
Compare to Dr. Scholl's® OneStep
Corn Removers active ingredient‡‡
Walgreens Corn RemoverSALICYLIC ACID STRIP
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||