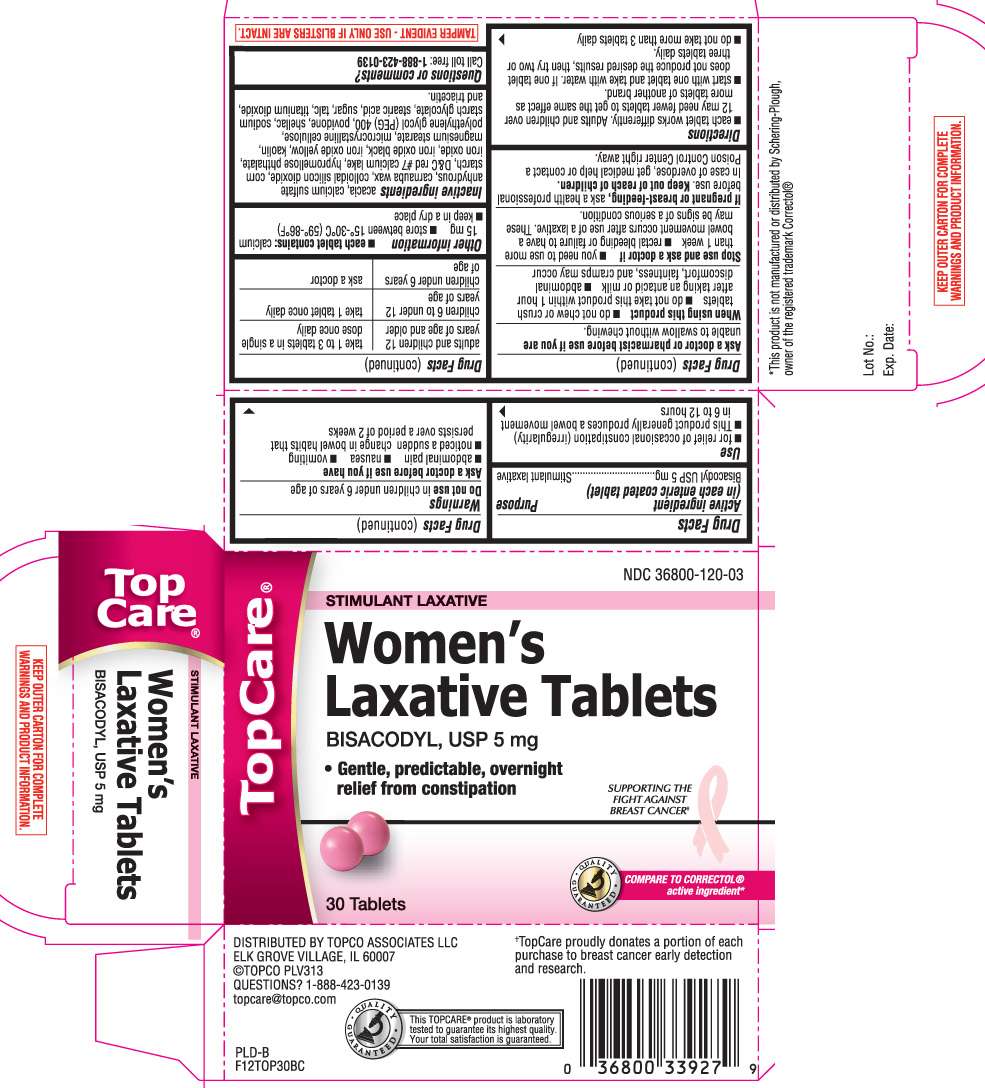
Womens Laxative
TOP CARE (Topco Associates LLC)
P and L Development of New York Corporation
Drug Facts
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active ingredient (in each enteric coated tablet)
- Purpose
- Use
- Warnings
- Directions
- Womens Laxative Other information
- Inactive ingredients
- Questions or comments?
- Principal Display Panel
- Product Label
FULL PRESCRIBING INFORMATION
Active ingredient (in each enteric coated tablet)
Bisacodyl USP 5 mg
Purpose
Stimulant laxative
Use
- for relief of occasional constipation (irregularity)
- this product generally produces a bowel movement in 6 to 12 hours
Warnings
Do not use
in children under 6 years of age.
Ask a doctor before use if you have
- abdominal pain
- nausea
- vomiting
- noticed a sudden change in bowel habits that persists over a period of 2 weeks
Ask a doctor or pharmacist before use if you are
unable to swallow without chewing.
When using this product
- do not chew or crush tablets
- do not take this product within 1 hour after taking an antacid or milk
- abdominal discomfort, faintness, and cramps may occur
Stop use and ask a doctor if
- you need to use more than 1 week
- rectal bleeding or failure to have a bowel movement occurs after use of a laxative. These maybe signs of serious condition.
If pregnant or breast-feeding,
ask a health professional before use.
Keep out of reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- each tablet works differently. Adults and children over 12 may need fewer tablets to get the same effect as more tablets of another brand.
- start with one tablet and take with water. If one tablet does not produce the desired results, then try two or three tablets daily.
- do not take more than 3 tablets daily
| adults and children 12 years of age and older | take 1 to 3 tablets in a single dose once daily |
| children 6 to under 12 years of age | take 1 tablet once daily |
| children under 6 years of age | ask a doctor |
Womens Laxative Other information
- each tablet contains: calcium 15 mg
- store between 15º-30ºC (59º-86ºF)
- keep in a dry place
Inactive ingredients
acacia, calcium sulfate anhydrous, carnauba wax, colloidal silicon dioxide, corn starch, D&C red #7 calcium lake, hypromellose phthalate, iron oxide, iron oxide black, iron oxide yellow, kaolin, magnesium stearate, microcrystalline cellulose, polyethylene glycol (PEG) 400, povidone, shellac, sodium starch glycolate, stearic acid, sugar, talc, titanium dioxide, and triacetin
Questions or comments?
Call toll free: 1-888-423-0139
Principal Display Panel
STIMULANT LAXATIVE
Women's Laxative Tablets
BISACODYL, USP 5 mg
- Gentle, predictable, overnight relief from constipation
Tablets
COMPARE TO CORRECTOL® active ingredient*
DISTRIBUTED BY TOPCO ASSOCIATES LLC
ELK GROVE VILLAGE, IL 60007
*This product is not manufactured or distributed by Schering-Plough, owner of the registered trademark Correctol®.
TAMPER EVIDENT- USE ONLY IF BLISTERS ARE INTACT.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCTS INFORMATION.
Product Label
Womens LaxativeBisacodyl TABLET, COATED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||