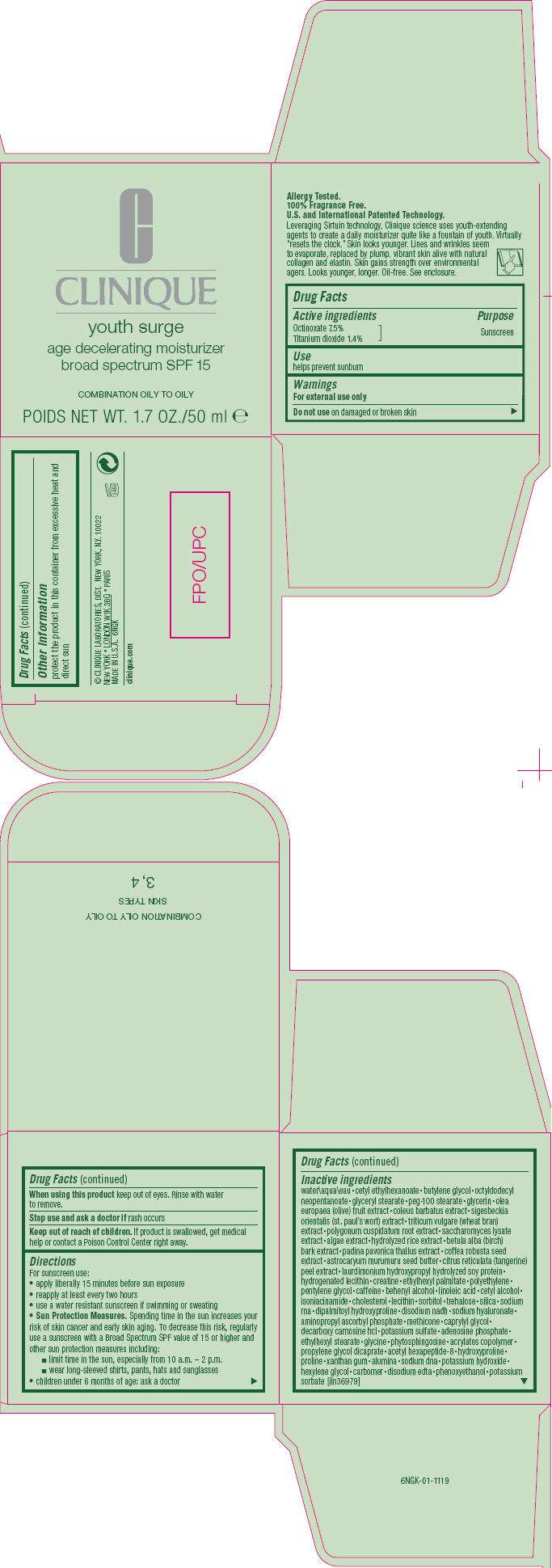
YOUTH SURGE
youth surge age decelerating moisturizer broad spectrum SPF 15
FULL PRESCRIBING INFORMATION: CONTENTS*
- Active ingredients
- Purpose
- Use
- Warnings
- Directions
- Inactive ingredients
- YOUTH SURGE Other information
- PRINCIPAL DISPLAY PANEL - 50 ml Carton
FULL PRESCRIBING INFORMATION
Drug Facts
Active ingredients
Octinoxate 7.5%
Titanium dioxide 1.4%
Purpose
Sunscreen
Use
helps prevent sunburn
Warnings
For external use only
Do not use on damaged or broken skin
When using this product keep out of eyes. Rinse with water to remove.
Stop use and ask a doctor if rash occurs
Keep out of reach of children. If product is swallowed, get medical help or contact a Poison Control Center right away.
Directions
For sunscreen use:
- apply liberally 15 minutes before sun exposure
- reapply at least every two hours
- use a water resistant sunscreen if swimming or sweating
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeved shirts, pants, hats and sunglasses
- children under 6 months of age: ask a doctor
Inactive ingredients
water • cetyl ethylhexanoate • butylene glycol • octyldodecyl neopentanoate • glyceryl stearate • peg-100 stearate • glycerin • olea europaea (olive) fruit extract • coleus barbatus extract • sigesbeckia orientalis (st. paul's wort) extract • triticum vulgare (wheat bran) extract • polygonum cuspidatum root extract • saccharomyces lysate extract • algae extract • hydrolyzed rice extract • betula alba (birch) bark extract • padina pavonica thallus extract • coffea robusta seed extract • astrocaryum murumuru seed butter • citrus reticulata (tangerine) peel extract • laurdimonium hydroxypropyl hydrolyzed soy protein • hydrogenated lecithin • creatine • ethylhexyl palmitate • polyethylene • pentylene glycol • caffeine • behenyl alcohol • linoleic acid • cetyl alcohol • isoniacinamide • cholesterol • lecithin • sorbitol • trehalose • silica • sodium rna • dipalmitoyl hydroxyproline • disodium nadh • sodium hyaluronate • aminopropyl ascorbyl phosphate • methicone • caprylyl glycol • decarboxy carnosine hcl • potassium sulfate • adenosine phosphate • ethylhexyl stearate • glycine • phytosphingosine • acrylates copolymer • propylene glycol dicaprate • acetyl hexapeptide-8 • hydroxyproline • proline • xanthan gum • alumina • sodium dna • potassium hydroxide • hexylene glycol • carbomer • disodium edta • phenoxyethanol • potassium sorbate [iln36979]
YOUTH SURGE Other information
protect the product in this container from excessive heat and direct sun
© CLINIQUE LABORATORIES, DIST. NEW YORK, N.Y. 10022
PRINCIPAL DISPLAY PANEL - 50 ml Carton
C
CLINIQUE
youth surge
age decelerating moisturizer
broad spectrum SPF 15
COMBINATION OILY TO OILY
POIDS NET WT. 1.7 OZ./50 ml e