Ziprasidone
HIGHLIGHTS OF PRESCRIBING INFORMATIONHIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Ziprasidone safely and effectively. See full prescribing information for ziprasidone. Ziprasidone HCl capsules Initial U.S. Approval: 2001 BOXED WARNING WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS See full prescribing information for complete boxed warning Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death compared to placebo treatment (5.1) Ziprasidone is not approved for elderly patients with dementia-related psychosis (5.1) INDICATIONS AND USAGE INDICATIONS AND USAGE Ziprasidone is an atypical antipsychotic. In choosing among treatments, prescribers should be aware of the capacity of ziprasidone to prolong the QT interval and may consider the use of other drugs first. (5.2)Ziprasidone is indicated as an oral formulation for the:Treatment of schizophrenia. (1.1) Adults: Efficacy was established in four 4-6 week trials and one maintenance trial in adults patients with schizophrenia. (14.1) Adults: Efficacy was established in two 3-week trials in adult patients with manic or mixed episodes. (14.2) Adults: Efficacy was established in one maintenance trial in adult patients. (14.2) DOSAGE AND ADMINISTRATION DOSAGE AND ADMINISTRATION Give oral doses with food. Schizophrenia: Initiate at 20 mg twice daily. Daily dosage may be adjusted up to 80 mg twice daily. Dose adjustments should occur at intervals of not less than 2 days. Safety and efficacy has been demonstrated in doses up to 100 mg twice daily. The lowest effective dose should be used. (2.1) Acute treatment of manic/mixed episodes of bipolar I disorder: Initiate at 40 mg twice daily. Increase to 60 mg or 80 mg twice daily on day 2 of treatment. Subsequent dose adjustments should be based on tolerability and efficacy within the range of 40-80 mg twice daily. (2.2) Maintenance treatment of bipolar I disorder as an adjunct to lithium or valproate. Continue treatment at the same dose on which the patient was initially stabilized, within the range of 40-80 mg twice daily. (2.2) DOSAGE FORMS AND STRENGTHS DOSAGE FORMS AND STRENGTHS Capsules: 20 mg, 40 mg, 60 mg, and 80 mg (3) CONTRAINDICATIONS CONTRAINDICATIONS Do not use in patients with a known history of QT prolongation. (4.1) Do not use in patients with recent acute myocardial infarction. (4.1) Do not use in patients with uncompensated heart failure. (4.1) Do not use in combination with other drugs that have demonstrated QT prolongation. (4.1) Do not use in patients with known hypersensitivity to ziprasidone. (4.2) WARNINGS AND PRECAUTIONS WARNINGS AND PRECAUTIONS QT Interval Prolongation: Ziprasidone use should be avoided in patients with bradycardia, hypokalemia or hypomagnesemia, congenital prolongation of the QT interval, or in combination with other drugs that have demonstrated QT prolongation. (5.2) Neuroleptic Malignant Syndrome (NMS): Potentially fatal symptom complex has been reported with antipsychotic drugs. Manage with immediate discontinuation of drug and close monitoring. (5.3) Tardive Dyskinesia: May develop acutely or chronically. (5.4) Hyperglycemia and Diabetes Mellitus (DM): Any patient treated with atypical antipsychotics should be monitored for symptoms of hyperglycemia including polydipsia, polyuria, polyhagia, and weakness. When starting treatment, patients with DM risk factors should undergo blood glucose testing before and during treatment. (5.5) Rash: Discontinue in patients who develop a rash without an identified cause. (5.6) Orthostatic Hypotension: Use with caution in patients with known cardiovascular or cerebrovascular disease. (5.7) Leukopenia, Neutropenia, and Agranulocytosis has been reported with antipsychotics. Patients with a pre-existing low white blood cell count (WBC) or a history of leukopenia/neutropenia should have their complete blood count (CBC) monitored frequently during the first few months of therapy and should discontinue ziprasidone at the first sign of a decline in WBC in the absence of other causative factors. (5.8) Seizures: Use cautiously in patients with a history of seizures or with conditions that lower seizure threshold. (5.9) Potential for Cognitive and Motor impairment: Patients should use caution when operating machinery. (5.12) Suicide: Closely supervise high-risk patients. (5.15) Side Effects ADVERSE REACTIONS Commonly observed adverse reactions (incidence greater than or equal to 5% and at least twice the incidence for placebo) were: Schizophrenia: Somnolence, respiratory tract infection. (6.1) Manic and Mixed Episodes Associated with Bipolar Disorder: Somnolence, extrapyramidal symptoms, dizziness, akathisia, abnormal vision, asthenia, vomiting. (6.1) To report SUSPECTED ADVERSE REACTIONS, call 1-800-438-1985 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.DRUG INTERACTIONS DRUG INTERACTIONS Ziprasidone should be used in combination with other drugs that have demonstrated QT prolongation. (4.1, 7.3) The absorption of ziprasidone is increased up to two-fold in the presence of food. (7.9) The full prescribing information contains additional drug interactions. (7) USE IN SPECIFIC POPULATIONS USE IN SPECIFIC POPULATIONS Pregnancy: Ziprasidone should be used during pregnancy only if the potential benefit justifies the potential risk. (8.1). Nursing Mothers: Breast feeding is not recommended. (8.3) Pediatric Use: Safety and effectiveness for pediatric patients has not been established. (8.4)
FULL PRESCRIBING INFORMATION: CONTENTS*
- FULL PRESCRIBING INFORMATION
- WARNING
- INDICATIONS & USAGE
- DOSAGE & ADMINISTRATION
- DOSAGE FORMS & STRENGTHS
- ZIPRASIDONE CONTRAINDICATIONS
- WARNINGS AND PRECAUTIONS
- ZIPRASIDONE ADVERSE REACTIONS
- DRUG INTERACTIONS
- USE IN SPECIFIC POPULATIONS
- DRUG ABUSE AND DEPENDENCE
- OVERDOSAGE
- ZIPRASIDONE DESCRIPTION
- CLINICAL PHARMACOLOGY
- NONCLINICAL TOXICOLOGY
- CLINICAL STUDIES
- HOW SUPPLIED
- STORAGE AND HANDLING
- PATIENT COUNSELING INFORMATION
- PATIENT SUMMARY OF INFORMATION ABOUT
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION
FULL PRESCRIBING INFORMATION
FULL PRESCRIBING INFORMATION
WARNING: INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS1 INDICATIONS AND USAGE
1.1 Schizophrenia
1.2 Bipolar I Disorder (Acute Mixed or Manic Episodes and Maintenance Treatment as an Adjunct to Lithium or Valproate)
2 DOSAGE AND ADMINISTRATION
2.1 Schizophrenia
2.2 Bipolar I Disorder (Acute Mixed or Manic Episodes and Maintenance Treatment as an Adjunct to Lithium or Valproate)
2.3 Dosing in Special Populations
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
4.1 QT Prolongation
4.2 Hypersensitivity
5 WARNINGS AND PRECAUTIONS
5.1 Increased Mortality in Elderly Patients with Dementia-Related Psychosis
5.2 QT Prolongation and Risk of Sudden Death
5.3 Neuroleptic Malignant Syndrome (NMS)
5.4 Tardive Dyskinesia
5.5 Hyperglycemia and Diabetes Mellitus
5.6 Rash
5.7 Orthostatic Hypotension
5.8 Leukopenia, Neutropenia, and Agranulocytosis
5.9 Seizures
5.10 Dysphagia
5.11 Hyperprolactinemia
5.12 Potential for Cognitive and Motor Impairment
5.13 Priapism
5.14 Body Temperature Regulation
5.15 Suicide
5.16 Patients with concomitant illnesses
5.17 Laboratory Tests
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Metabolic Pathway
7.2 In Vitro Studies
7.3 Pharmacodynamic Interactions
7.4 Pharmacokinetic Interactions
7.5 Lithium
7.6 Oral Contraceptives
7.7 Dextromethorphan
7.8 Valproate
7.9 Other Concomitant Drug Therapy
7.10 Food Interaction
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Labor and Delivery
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
8.8 Age and Gender Effects
8.9 Smoking
9 DRUG ABUSE AND DEPENDENCE
9.3 Dependence
10 OVERDOSAGE
10.1 Human Experience
10.2 Management of Overdosage
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Schizophrenia
14.2 Bipolar I Disorder (Acute Mixed or Manic Episodes and Maintenance Treatment as an Adjunct to Lithium or Valproate)
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
17.1 Administration with Food
17.2 QTc Prolongation
17.3 FDA-Approved Patient Labeling
WARNING
INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSISElderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Analyses of seventeen placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies may be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. Ziprasidone is not approved for the treatment of patients with Dementia-Related Psychosis [see Warnings and Precautions (5.1)]).
INDICATIONS & USAGE
Warnings and Precautions (5.2)]. Prolongation of the QTc interval is associated in some other drugs with the ability to cause torsade de pointes-type arrhythmia, a potentially fatal polymorphic ventricular tachycardia, and sudden death. In many cases this would lead to the conclusion that other drugs should be tried first. Whether ziprasidone will cause torsade de pointes or increase the rate of sudden death is not yet known [seeWarnings and Precautions (5.2)]1.1 Schizophrenia
Clinical Studies (14.1)].
1.2 Bipolar I Disorder (Acute Mixed or Manic Episodes and Maintenance Treatment as an Adjunct to Lithium or Valproate)
Clinical Studies (14.2)].
Clinical Studies (14.2)].
DOSAGE & ADMINISTRATION
2.1 SchizophreniaDose Selection
Clinical Studies (14.1)].
Maintenance Treatment
Clinical Studies (14.1)]. No additional benefit was demonstrated for doses above 20 mg twice daily. Patients should be periodically reassessed to determine the need for maintenance treatment.
2.2 Bipolar I Disorder (Acute Mixed or Manic Episodes and Maintenance Treatment as an Adjunct to Lithium or Valproate)
Acute Treatment of Manic or Mixed Episodes
Clinical Studies (14.2)].
Maintenance Treatment (as an adjunct to lithium or valproate)
Clinical Studies (14.2)]
2.3 Dosing in Special Populations
DOSAGE FORMS & STRENGTHS
Capsule Strength (mg) Imprint 20 2001 40 2002 60 2003 80 2004
ZIPRASIDONE CONTRAINDICATIONS
4.1 QT ProlongationWarnings and Precautions (5.2)].
4.2 Hypersensitivity
WARNINGS AND PRECAUTIONS
5.1 Increased Mortality in Elderly Patients with Dementia-Related PsychosisElderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Ziprasidone is not approved for the treatment of dementia-related psychosis. [see Boxed Warning]
5.2 QT Prolongation and Risk of Sudden Death
Contraindications (4.1),Drug Interactions (7.4)]. Additionally, clinicians should be alert to the identification of other drugs that have been consistently observed to prolong the QTc interval. Such drugs should not be prescribed with ziprasidone. Ziprasidone should also be avoided in patients with congenital long QT syndrome and in patients with a history of cardiac arrhythmias [seeContraindications (4)].
Adverse Reactions (6.2)].
Indications and Usage (1)].
5.3 Neuroleptic Malignant Syndrome (NMS)
5.4 Tardive Dyskinesia
5.5 Hyperglycemia and Diabetes Mellitus
5.6 Rash
5.7 Orthostatic Hypotension
5.8 Leukopenia, Neutropenia, and Agranulocytosis
5.9 Seizures
5.10 Dysphagia
Boxed Warning].
5.11 Hyperprolactinemia
Nonclinical Toxicology (13.1)]. Tissue culture experiments indicate that approximately one-third of human breast cancers are prolactin-dependent in vitro, a factor of potential importance if the prescription of these drugs is contemplated in patient with previously detected breast cancer. Although disturbances such as galactorrhea, amenorrhea, gynecomastia, and impotence have been reported with prolactin-elevating compounds, the clinical significance of elevated serum prolactin levels is unknown for most patients. Neither clinical studies nor epidemiologic studies conducted to date have shown an association between chronic administration of this class of drugs and tumorigenesis in humans; the available evidence is considered too limited to be conclusive at this time.
5.12 Potential for Cognitive and Motor Impairment
5.13 Priapism
5.14 Body Temperature Regulation
5.15 Suicide
5.16 Patients with concomitant illnesses
Use in Specific Populations (8.6),( 8.7)]
Warnings and Precautions (5.2),(5.7)]
5.17 Laboratory Tests
Warnings and Precautions (5.2)]
ZIPRASIDONE ADVERSE REACTIONS
6.1 Clinical Trials ExperienceAdverse Findings Observed in Short-Term, Placebo-Controlled Trials with Oral Ziprasidone
Table 1)
Table 2)
SCHIZOPHRENIA
Adverse Reactions Associated with Discontinuation of Treatment in Short-Term, Placebo-Controlled Trials of Oral Ziprasidone
Warnings and Precautions (5.6)].
Adverse Reactions Occurring at an Incidence of 2% or More Among Ziprasidone-Treated Patients in Short-Term, Oral, Placebo-Controlled Trials
Table 1: Treatment-Emergent Adverse Reaction Incidence In Short-Term Oral Placebo-Controlled TrialsSchizophrenia
Percentage of Patients
Reporting ReactionBody System/Adverse Reaction Ziprasidone
(N=702) Placebo
(N=273) *
Body as a Whole Cardiovascular Digestive Nervous *Respiratory Skin and Appendages Special Senses
Dose Dependency of Adverse Reactions in Short-Term, Fixed-Dose, Placebo-Controlled Trials
Extrapyramidal Symptoms (EPS)- The incidence of reported EPS (which included the adverse reaction terms extrapyramidal syndrome, hypertonia, dystonia, dyskinesia, hypokinesia, tremor, paralysis and twitching) for ziprasidone-treated patients in the short-term, placebo-controlled schizophrenia trials was 14% vs. 8% for placebo. Objectively collected data from those trials on the Simpson-Angus Rating Scale (for EPS) and the Barnes Akathisia Scale (for akathisia) did not generally show a difference between ziprasidone and placebo.
Dystonia- Class Effect: Symptoms of dystonia, prolonged abnormal contractions of muscle groups, may occur in susceptible individuals during the first few days of treatment. Dystonic symptoms include: spasm of the neck muscles, sometimes progressing to tightness of the throat, swallowing difficulty, difficulty breathing, and/or protrusion of the tongue. While these symptoms can occur at low doses, they occur more frequently and with greater severity with high potency and at higher doses of first generation antipsychotic drugs. An elevated risk of acute dystonia is observed in males and younger age groups.
Vital Sign Changes- Ziprasidone is associated with orthostatic hypotension [seeWarnings and Precautions (5.7)
Weight Gain- The proportions of patients meeting a weight gain criterion of greater than or equal to 7% of body weight were compared in a pool of four 4- and 6-week placebo-controlled schizophrenia clinical trials, revealing a statistically significantly greater incidence of weight gain for ziprasidone (10%) compared to placebo (4%). A median weight gain of 0.5 kg was observed in ziprasidone patients compared to no median weight change in placebo patients. In this set of clinical trials, weight gain was reported as an adverse reaction in 0.4% and 0.4% of ziprasidone and placebo patients, respectively. During long-term therapy with ziprasidone, a categorization of patients at baseline on the basis of body mass index (BMI) revealed the greatest mean weight gain and highest incidence of clinically significant weight gain (>7% of body weight) in patients with low BMI (<23) compared to normal (23-27) or overweight patients (>27). There was a mean weight gain of 1.4 kg for those patients with a "low" baseline BMI, no mean change for patients with a "normal" BMI, and a 1.3 kg mean weight loss for patients who entered the program with a "high" BMI.
ECG Changes- Ziprasidone is associated with an increase in the QTc interval [seeWarnings and Precautions (5.2)]. In the schizophrenia trials, ziprasidone was associated with a mean increase in heart rate of 1.4 beats per minute compared to a 0.2 beats per minute decrease among placebo patients.
Other Adverse Reactions Observed During the Premarketing Evaluation of Oral Ziprasidone
ADVERSE REACTIONSsection reported by patients treated with ziprasidone in schizophrenia trials at multiple doses >4 mg/day within the database of 3834 patients. All reported reactions are included except those already listed in Table 1 or elsewhere in labeling, those reaction terms that were so general as to be uninformative, reactions reported only once and that did not have a substantial probability of being acutely life-threatening, reactions that are part of the illness being treated or are otherwise common as background reactions, and reactions considered unlikely to be drug-related. It is important to emphasize that, although the reactions reported occurred during treatment with ziprasidone, they were not necessarily caused by it.
Body as a Whole
Cardiovascular System
Digestive System
Endocrine
Hemic and Lymphatic System
Metabolic and Nutritional Disorders
Musculoskeletal System
Nervous System
Respiratory System
Skin and Appendages
Special Senses
Urogenital System
BIPOLAR DISORDER
Acute Treatment of Manic or Mixed Episodes
Adverse Reactions Associated with Discontinuation of Treatment in Short Term, Placebo-Controlled Trials
Adverse Reactions Occurring at an Incidence of 2% or More Among Ziprasidone-Treated Patients in Short-Term, Oral, Placebo-Controlled Trials
Table 2: Treatment-Emergent Adverse Reactions Incidence In Short-Term Oral Placebo-Controlled TrialsManic and Mixed Episodes Associated with Bipolar Disorder
Percentage of Patients
Reporting ReactionBody System/Adverse Reaction Ziprasidone
(N=279) Placebo
(N=136) *
Body as a Whole Cardiovascular Digestive Musculoskeletal Nervous *Respiratory Skin and Appendages Special Senses
Weight Gain-During a 6-month placebo-controlled bipolar maintenance study in adults with ziprasidone as an adjunct to lithium or valproate, the incidence of clinically significant weight gain (greater than or equal to 7% of body weight) during the double-blind period was 5.6% for both ziprasidone and placebo treatment groups who completed the 6 months of observation for relapse. Interpretation of these findings should take into consideration that only patients who adequately tolerated ziprasidone entered the maintenance phase of this study, and there were substantial dropouts by the 6 month endpoint.
6.2 Postmarketing Experience
Warnings and Precautions (5.2)]; Digestive System Disorder: Swollen Tongue; Reproductive System and Breast Disorders: Galactorrhea, priapism; Nervous System Disorders: Facial Droop, neuroleptic malignant syndrome, serotonin syndrome (alone or in combination with serotonergic medicinal products), tardive dyskinesia; Psychiatric Disorders: Insomnia, mania/hypomania; Skin and subcutaneous Tissue Disorders: Allergic reaction (such as allergic dermatitis, angioedema, orofacial edema, urticaria), rash; Urogenital System Disorders: Enuresis, urinary incontinence; Vascular Disorders: Postural hypotension, syncope.
DRUG INTERACTIONS
7.1 Metabolic Pathway
7.2 In Vitro Studies
Clinical Pharmacology (12.3)].
7.3 Pharmacodynamic Interactions
Contraindications (4.1)].
7.4 Pharmacokinetic Interactions
Carbamazepine
Ketoconazole
Cimetidine
Antacid
7.5 Lithium
7.6 Oral Contraceptives
7.7 Dextromethorphan
7.8 Valproate
7.9 Other Concomitant Drug Therapy
7.10 Food Interaction
Clinical Pharmacology (12.3)].
USE IN SPECIFIC POPULATIONS
8.1 Pregnancy8.2 Labor and Delivery
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
8.8 Age and Gender Effects
8.9 Smoking
DRUG ABUSE AND DEPENDENCE
9.3 DependenceOVERDOSAGE
10.1 Human ExperienceAdverse Reactions (6.2)]
10.2 Management of Overdosage
ZIPRASIDONE DESCRIPTION
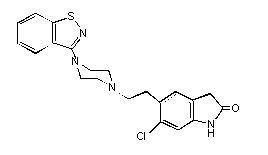
CLINICAL PHARMACOLOGY
12.1 Mechanism of Action12.2 Pharmacodynamics
12.3 Pharmacokinetics
Oral Pharmacokinetics
Absorption:Ziprasidone is well absorbed after oral administration, reaching peak plasma concentrations in 6 to 8 hours. The absolute bioavailability of a 20 mg dose under fed conditions is approximately 60%. The absorption of ziprasidone is increased up to two-fold in the presence of food.
Distribution:Ziprasidone has a mean apparent volume of distribution of 1.5 L/kg. It is greater than 99% bound to plasma proteins, binding primarily to albumin andglycoprotein. The in vitro plasma protein binding of ziprasidone was not altered by warfarin or propranolol, two highly protein-bound drugs, nor did ziprasidone alter the binding of these drugs in human plasma. Thus, the potential for drug interactions with ziprasidone due to displacement is minimal.
Metabolism and Elimination:Ziprasidone is extensively metabolized after oral administration with only a small amount excreted in the urine (<1%) or feces (<4%) as unchanged drug. Ziprasidone is primarily cleared via three metabolic routes to yield four major circulating metabolites, benzisothiazole (BITP) sulphoxide, BITP-sulphone, ziprasidone sulphoxide, and S-methyl-dihydroziprasidone. Approximately 20% of the dose is excreted in the urine, with approximately 66% being eliminated in the feces. Unchanged ziprasidone represents about 44% of total drug-related material in serum. In vitro studies using human liver subcellular fractions indicate that S-methyldihydroziprasidone is generated in two steps. These studies indicate that the reduction reaction is mediated primarily by chemical reduction by glutathione as well as by enzymatic reduction by aldehyde oxidase and the subsequent methylation is mediated by thiol methyltransferase. In vitro studies using human liver microsomes and recombinant enzymes indicate that CYP3A4 is the major CYP contributing to the oxidative metabolism of ziprasidone. CYP1A2 may contribute to a much lesser extent. Based on in vivo abundance of excretory metabolites, less than one-third of ziprasidone metabolic clearance is mediated by cytochrome P450 catalyzed oxidation and approximately two-thirds via reduction. There are no known clinically relevant inhibitors or inducers of aldehyde oxidase.
NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of FertilityCarcinogenesis
Warnings and Precautions (5.11)].
Mutagenesis
Impairment of Fertility
CLINICAL STUDIES
14.1 SchizophreniaThe results of the oral ziprasidone trials in schizophrenia follow:
14.2 Bipolar I Disorder (Acute Mixed or Manic Episodes and Maintenance Treatment as an Adjunct to Lithium or Valproate)
Acute Manic and Mixed Episodes Associated with Bipolar I Disorder
Maintenance Therapy
HOW SUPPLIED
Ziprasidone Capsules
Package Configuration Capsule Strength (mg) NDC Code Imprint
STORAGE AND HANDLING
PATIENT COUNSELING INFORMATION
FDA-Approved Patient Labeling (17.3).17.1 Administration with Food
Drug Interactions (7.8)andClinical Pharmacology (12.3)].
17.2 QTc Prolongation
Contraindications (4.1)andWarnings and Precautions (5.2)].
Warnings and Precautions (5.2)].
17.3 FDA-Approved Patient Labeling

PATIENT SUMMARY OF INFORMATION ABOUT
Ziprasidone Capsules(ziprasidone HCl)
Information for patients taking ziprasidone or their caregivers
What Is ziprasidone?
Who Should Take ziprasidone?
What is the most important safety information I should know about ziprasidone?
Ziprasidone is not approved for the treatment of patients with dementia-related psychosis. Elderly patients with a diagnosis of psychosis related to dementia treated with antipsychotics are at an increased risk of death when compared to patients who are treated with placebo (a sugar pill).
Your risk of dangerous changes in heart rhythm can be increased if you are taking certain other medicines and if you already have certain abnormal heart conditions. Therefore, it is important to tell your doctor about any other medicines that you take, including non-prescription medicines, supplements, and herbal medicines. You must also tell your doctor about any heart problems you have or have had.
Who should NOT take ziprasidone?
What To Tell Your Doctor Before You Start ziprasidone
Ziprasidone And Other Medicines
How To Take ziprasidone
Possible Side Effects
IMMEDIATELYif you:
What To Do For An Overdose
Other Important Safety Information
immediatelyif you take more than the amount of ziprasidone prescribed by your doctor.
Keep ziprasidone and all medicines out of the reach of children.
How To Store ziprasidone
For More Information About ziprasidone
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL SECTION

ZiprasidoneZIPRASIDONE HYDROCHLORIDE CAPSULE
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PLEASE, BE CAREFUL!
Be sure to consult your doctor before taking any medication!